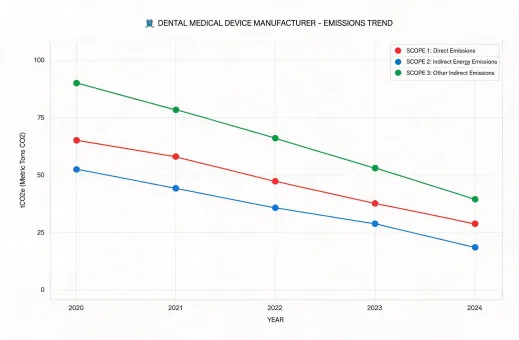
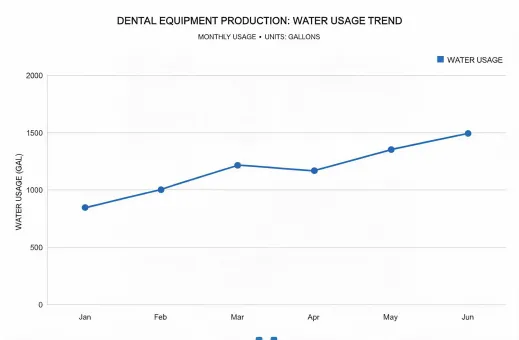

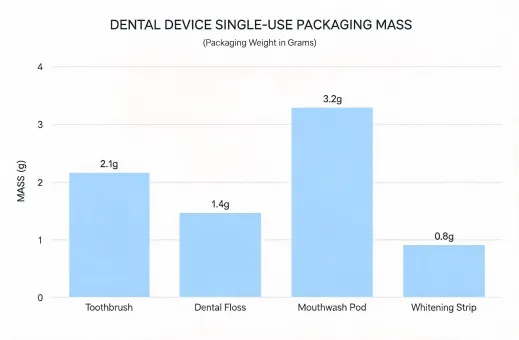

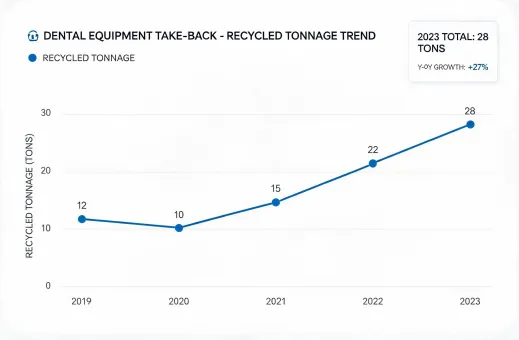
Reporting under SASB Medical Equipment & Supplies, GRI, TCFD, and practical clinic-level take-back expectations.






Dental buyers increasingly ask how equipment refreshes affect packaging waste, power draw, spare-parts logistics, device take-back, and long-term maintenance. The disclosure pack organizes those details without making claims that outrun the underlying data.
Environmental claims and materials information are connected to technical documentation rather than isolated marketing statements.
Manufacturing location, shipping mode, and equipment weight become part of procurement scenario planning for international dental groups.
Software patching, sleep-mode behavior, remote diagnostics, and backup retention are reviewed together so connected dental workflows remain resilient.
Use it to support RFP responses, value-analysis review, and multi-site dental equipment replacement planning.
Get the Data